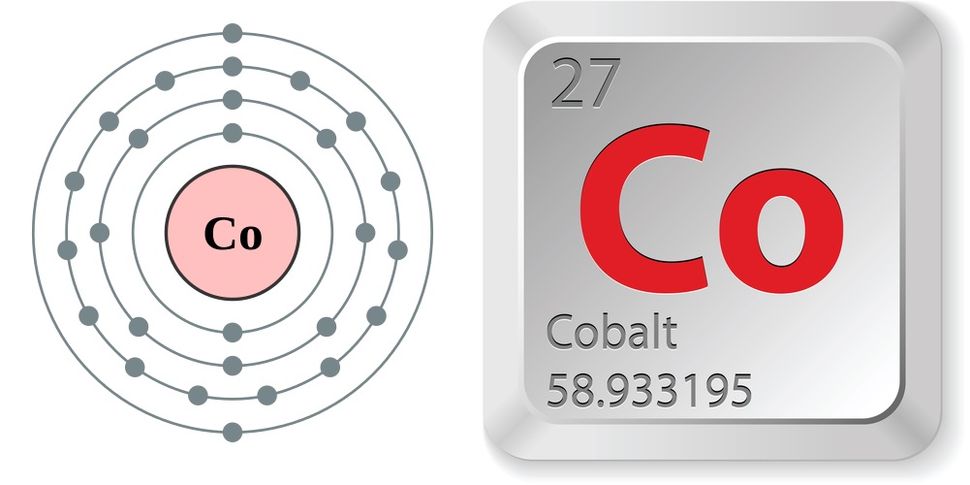 
Cobalt Compounds:Īlthough states of +4, +1, 0, and 1 are known, cobalt almost invariably exhibits a +2 or +3 oxidation state in its compounds. Larger chunks are rather inert in air, although substantial oxidation happens above 300 ☌ (570 ☏). The most effective matrix for cemented carbides is cobalt.Ĭobalt that has been finely split ignites naturally. Hard-facing alloys, tool steels, low-expansion alloys (for glass-to-metal sealing), and constant-modulus (elastic) alloys are further applications for cobalt (for precision hairsprings). Large volumes are employed for superalloys that are used close to their melting points and alloys that maintain their characteristics at high temperatures (where steels would become too soft). Magnetic alloys, including Alnicos for permanent magnets, account for a sizable portion of global production. Special alloys are where most of the cobalt produced is used. Additionally, it has been employed as a radioactive tracer in biology and industry, sterilization research, and cancer therapy. X-rays and radium alpha rays have been replaced with cobalt-60 gamma radiation in the inspection of industrial materials to identify interior structures, defects, or foreign items. The longest-lived manmade radioactive isotope, cobalt-60 (with a half-life of 5.3 years), is created by neutron irradiation in a nuclear reactor from the stable isotope cobalt-59, which makes up all naturally occurring cobalt. At high temperatures, oxygen and water vapor also attack cobalt, causing cobaltous oxide, or CoO (in the +2 state with the metal), to be created. It slowly dissolves in weak mineral acids, does not interact with hydrogen or nitrogen immediately, but will combine with carbon, phosphorus, or sulfur when heated. One of the three metals that exhibit ferromagnetism at normal temperature is cobalt. It can be used in situations where magnetic qualities are required at high temperatures since it is ferromagnetic up to 1,121 ☌ (2,050 ☏), which is the highest known Curie point of any metal or alloy. There are two known allotropes: the face-centered cubic, which is stable at high temperatures, and the hexagonal close-packed structure, which is stable below 417 ☌ (783 ☏). Cobalt-60, which has a half-life of 5.3 years and is frequently employed in medicinal applications, is the most common.Ĭobalt, in general, is a silver-white with a slightly blue hue, polished metal. At temperatures below 417 ☌ (783 ☏), it exhibits a hexagonal closed pack crystal structure and contains 12 radioactive isotopes. Boiling Point is recorded at 5612☏ or 2870☌. The Classification includes Crystallized Transition Metal Hexagonal close-packed structure. Its sign is “Co.” Its Atomic Weight is 27. Cobalt’s main characteristics are listed below: It has excellent conduction qualities and a high strength. It has a high melting point and endures well in hot conditions. The ferromagnetic metal cobalt is hard but fragile. 
It is a member of the periodic table’s first-row transition series and cannot be found naturally in its purest form rather, it can only be found in combinations with other elements. Cobalt is unaffected by water and stable in air, although weak acids progressively corrode it. Chemically, the element is active, creating several compounds. Its physical characteristics are comparable to those of nickel and iron. It belongs to the periodic table’s group VIII. Cobalt is a brittle, silver-white, hard, ferromagnetic, and glossy element. 
In nature, cobalt (Co) is a rare metal element that makes up 0.001% of the crust of the planet. In many uses, it is typically mixed with iron to create various alloys. Cobalt is an ore that, when properly mined and polished, yields a beautiful, bluish-white metal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
